Informing on science news in the world
Provided by AGPWatching titania break down methanol, one molecule at a time
FAYETTEVILLE, GA, UNITED STATES, May 11, 2026 /EINPresswire.com/ -- Titanium dioxide (TiO₂) is a promising photocatalyst for converting methanol into cleaner fuels and chemicals, but the molecular steps of this reaction remain poorly understood. Using high-resolution photoelectron spectroscopy of cryogenically cooled gas-phase clusters, scientists have now captured how a single TiO₂ molecule splits methanol. The work reveals a dissociative adduct where methanol breaks apart upon binding, and measures the electron affinity (EA) of the resulting complex. These findings provide fundamental insights into methanol photooxidation, a reaction relevant to renewable energy and pollution control. Understanding this process at the single-molecule level helps bridge the gap between gas-phase model systems and real-world catalytic surfaces.
Methanol splitting on Titanium dioxide (TiO₂) surfaces plays a key role in photocatalytic hydrogen production and fuel cell technologies. However, the reaction occurs primarily at rare defect sites such as steps, edges, and vacancies on the bulk material, making direct experimental observation extremely difficult. Gas-phase metal oxide clusters offer a powerful alternative, allowing researchers to isolate and study reactive intermediates without the complexity of a full surface. These clusters can be formed in laser-ablation sources and characterized with high precision, controlling factors like particle size and ion charge that affect reactivity. Based on these challenges, there is a clear need for in-depth spectroscopic investigation of how individual TiO₂ molecules interact with methanol at the molecular level.
Researchers at the University of California, Berkeley, report (DOI: 10.1063/1674-0068/cjcp2510163) in the Chinese Journal of Chemical Physics (published online October 16, 2025) the first high-resolution photoelectron spectra of the TiO₂–methanol complex. Using cryogenically cooled anions and slow electron velocity-map imaging (cryo-SEVI), the team visualized how a single TiO₂ molecule splits methanol. The work yields precise measurements of electron affinity and vibrational frequencies that illuminate the reaction pathway, offering a molecular-scale view of a process central to photocatalytic energy conversion.
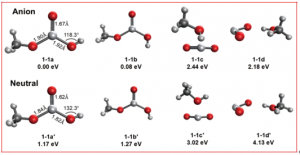
The team created negatively charged TiO₂–methanol clusters in a laser-ablation source, then cooled them to about 10 Kelvin in an ion trap before detaching electrons with tunable laser light. The resulting photoelectron spectra showed over 40 distinct features, revealing the electron affinity of the neutral TiO₂CH₃OH complex as 1.2152 eV — roughly 0.4 eV lower than that of bare TiO₂. This shift indicates that neutral TiO₂ reacts more exothermically with methanol than its anionic counterpart, pointing to a higher reactivity of the Ti(IV) oxidation state compared to Ti(III).
Most spectral peaks matched calculations for a dissociative adduct called cis-CH₃OTi(O)OH, where methanol’s O–H and C–O bonds have rearranged around the titanium center. A surprising set of weaker peaks, however, could not be explained by standard Franck-Condon (FC) simulations. The team traced these forbidden transitions to Herzberg-Teller (HT) coupling involving an excited electronic state of the anion — a subtle quantum mechanical effect rarely observed in photoelectron spectroscopy. The two types of transitions also showed distinct photoelectron angular distributions (PADs), with allowed transitions displaying positive anisotropy while forbidden ones appeared nearly isotropic near threshold. This discovery highlights how excited-state interactions can shape spectra in unexpected ways.
“Watching a single TiO₂ molecule split methanol gives us a bottom-up view of a reaction that happens constantly on catalyst surfaces but is nearly impossible to track directly,” the authors stated in this work, “The fact that the neutral titanium center, with its +4 oxidation state, binds methanol much more strongly than the anionic +3 state tells us that electron holes are key drivers of this chemistry. That aligns beautifully with what happens on real TiO₂ surfaces during photocatalysis, where light creates electron-hole pairs to power reactions. Gas-phase clusters let us isolate these effects without the complexity of a full surface.”
These findings help explain why TiO₂ is such an effective photocatalyst for breaking down organic molecules. The lower electron affinity of the methanol-split complex means neutral TiO₂ — the state created when light generates a hole — is more reactive toward methanol than the reduced form. This principle directly informs the design of better photocatalysts: strategies that stabilize the Ti(IV) oxidation state or promote hole formation could boost catalytic efficiency. Moreover, the gas-phase cluster approach demonstrated here can be extended to study other small-molecule activations, including water splitting and carbon dioxide reduction, offering a molecular-scale toolkit for developing next-generation energy conversion materials.
References
DOI
10.1063/1674-0068/cjcp2510163
Original Source URL
https://dx.doi.org/10.1063/1674-0068/cjcp2510163
Funding Information
This research was funded by the Air Force Office of Scientific Research (AFOSR) under Grant (No.FA9550-23-1-0545).
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.